Indicate whether the depicted vibration is active or inactive in the IR spectrum.
Question:
Indicate whether the depicted vibration is active or inactive in the IR spectrum.
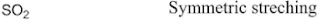 |
IR Active:
Some connections completely block infrared light, while others absorb it weakly. The periodic shift in the molecule's dipole moment requires a vibrational mode to absorb infrared light. According to some, these vibrations are infrared active.
Answer and Explanation: 1
Become a Study.com member to unlock this answer! Create your account
View this answerGiven Data
{eq}{\rm{S}}{{\rm{O}}_{\rm{2}}} {/eq} has symmetric stretching
Sulfur and oxygen are the two types of atoms in this molecule, and...
See full answer below.
Ask a question
Our experts can answer your tough homework and study questions.
Ask a question Ask a questionSearch Answers
Learn more about this topic:

from
Chapter 8 / Lesson 10Infrared energy has a longer wavelength than the visible spectrum. Explore how infrared spectroscopy (IR) is used to interpret infrared energy and create an identifiable spectrum and discover its applications in forensic science and homeland security.
Related to this Question
- Indicate whether the indicated vibration is active or inactive in the IR spectrum.
- Tell if the depicted vibration is active or inactive in the IR spectrum.
- Indicate whether the stated vibration is active or inactive in the IR spectrum.
- Indicate whether the mentioned vibration is active or inactive in the IR spectrum.
- Tell if the mentioned vibration is active or inactive in the IR spectrum.
- Determine the number of IR-active vibrations for the stated molecule. CHCl3
- Determine the number of IR-active vibrations for the depicted molecule. CH2Cl2
- A strong signal at 1700 cm^{-1} in an IR spectrum indicates the presence of a(n) a) Alcohol b) ether c) carbonyl d) Aminel
- Determine the number of IR-active vibrations for the indicated molecule. CCl4
- Identify the absorption in the IR spectrum of benzoin that indicates the benzoin condensation has occurred.
- Determine the number of IR-active vibrations for the depicted molecule. N(CH3)3 (assume C3v symmetry)
- The vibration of which bond gives an IR absorption that distinguishes between the following two compounds: CH_3CH_2CH_2OH and CH_3CH_2OCH_3. A. O-H B. C-O C. C-C D. C-H What is the approximate wavenum
- Classify the following as optically active or optically inactive, a solution of 1-isohexane.
- Classify the following as optically active or optically inactive, a solution of (S)-2-isohexane.
- Determine the number of IR-active vibrations for the given molecule. F2O
- Determine the number of IR-active vibrations for the given molecule. CH4
- Indicate all diagnostically relevant peaks expected for limonene and which bond stretch/vibration is responsible. Bond: Predicted Absorption cm^-1:
- Which of the following functional group exhibits the lowest stretching vibration frequency? (D = 2H) A) sp3 C-H B) sp2 C-H C) sp3 C-D D) sp2 C-D
- What functional group(s) might the molecules contain if they show IR absorption at the given frequencies? 3500 cm^(-1) and 1735 cm^(-1)
- At what approximate position(s) will the depicted compound show IR absorption?
- Identify all compounds that are likely to show a strong peak at about 1600-1800 cm^-1 in their IR spectra. (Hint: which functional group(s) would you be looking for?) 1) Sucralose 2) Retinol 3) Cholesterol 4) Allethrin 5) Aflatoxin 6) DDT
- Sketch an active vibrational mode of : o)CO2
- Sketch an active vibrational mode of : o)H2O
- For the following compounds and the indicated vibration, indicate if the vibrational transition will result in an observed (IR - active) absorption band. Explain. a. cis - 1,2-dichloroethene, C=C stretch. b. trans - 1,2-dichloroethene, symmetric C-Cl str
- The vibrational modes of XeF2 are at 555, 515, and 213 cm-1, but only two are IR active. Explain why this is consistent with XeF2 having a linear structure?
- What is Nujol (look it up online)? Where (what wavenumbers) does it absorb IR radiation (see attached IR)?
- At what approximate position(s) might the indicated compound show IR absorption?
- What Does it mean to be optically active? and if you are not optically active does it prevent you fromoxidizing the molecule?
- Determine the number of IR-active vibrations for benzene, C_6H_6. Does it agree with the material in the text?
- Sketch an inactive vibrational mode of : o)CO2
- What are the characteristic functional groups you should see in an infrared spectrum for Spandex?
- Propose structures for compounds that meet the following descriptions: (a) An optically active compound C_5H_10O with an IR absorption at 1730 cm^-1. (b) A non-optically active compound C_5H_9N with an IR absorption at 2215 cm^-1.
- Select the region of the IR spectrum where triphenylmethanol will NOT exhibit bands. 2300-2400 cm^(-1) 1750-1950 cm^(-1) 3400-3500 cm^(-1) 3000-3100 cm^(-1)
- In the IR spectrum, changes in which functional groups would indicate successful epoxidation?
- What kind(s) of functional groups might the compound contain if they show the mentioned IR absorption? 1670 cm-1
- The compound shows IR absorption for which position
- The compound shows IR absorption at which position
- Deduce the number of normal mode vibrations observed for the given molecule. BF3
- Choose the compou[{Image src='img45680210049494192156.jpg' alt='' caption=''}]nd that matches the IR spectrum shown. Choose the compound that matches the IR spectrum shown. Spectrum [5] 100 E 50 4 0 4
- What key peak(s) in an IR spectrum can allow you to distinguish a saturated triglyceride from an unsaturated one?
- The compound show IR absorptions at which approximate position
- At what approximate position(s) might the given compound show IR absorption?
- The gaseous N2O IR spectrum exhibits three prominent absorption bands at 2224 cm^-1, 1285 cm-1, and 2089 cm-1, along with two relatively weaker bands at 2563 cm-1 and 2798 cm-1. While N2O is known to be a linear molecule, the specific arrangement between
- Deduce the identity of the following compound from the data given. C_{4}H_{8}O, gave absorption bands at 1720 and 2700 cm^{-1}, but not at 3300 cm^{-1} in the IR spectrum; ^{13}\textrm{C} NMR: 4 signals
- At what approximate position(s) might the mentioned compound show IR absorption?
- What functional group(s) might the molecules contain if they show IR absorption at the frequency 2210 cm^(-1)?
- Where in the IR spectrum would you expect the following molecule to absorb?
- Where in the IR spectrum would you expect the following molecule below to absorb?
- Where in the IR spectrum would you expect the given molecule to absorb?
- Ongoing from Fe(CO)_5 to Fe(CO)_3(PPh_3)_2, absorptions in the IR spectrum at 2025
- Compound (C_5H_8O) has a strong IR absorption band at 1745 cm^-1 The broad-band proton decoupled,3C spectrum of this compound shows three signals: at 6 220 (C), 23 (CH2), and 38 (CH2). Propose a struc
- What are the characteristic absorption regions for carbonyl groups? Are these absorptions a result of stretching vibrations or bending vibrations?
- In IR, why does asymmetric stretch need more energy than a symmetric stretch?
- List three structural features and corresponding absorption ranges that can be used to identify citral by IR spectroscopy.?
- Compute the approximate position for IR absorption
- Determine the number of normal mode vibrations observed for the given molecule. CCl4
- What functional group(s) might the molecule contain if it shows IR absorption at the frequency 1715 cm^(-1)?
- 4) What component of the caffeine structure allows it to be classified as an alkaloid? 5) What bond vibrations and stretching frequencies should you observe in the IR spectrum of caffeine? 6) A 12 f...
- What functional group(s) might the molecule contain if it shows IR absorption at the frequency 1540 cm^(-1)?
- Which of the following functional group exhibits the lowest stretching vibration frequency? (D = ^2H) A) sp^3 C-H. B) sp^2 C-H. C) sp^3 C-D. D) sp^2 C-D.
- Indicate if the depicted expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d/dpie tanpie
- For each group of IR frequencies listed below, suggest the functional group that is present.
- What functional group(s) might the molecule contain if it shows IR absorption at the given frequencies? 1720 cm^(-1) and 2500-3100 cm^(-1)
- At what approximate positions might this compound show IR absorptions? [{Image src='irr4889887586621820989.jpg' alt='IRR' caption=''}]
- Determine the position for following compound at which it show IR absorptions?
- Determine the number of IR-active vibrations for the following molecule. Determine their symmetry first. Hydrogen peroxide, H_2O_2
- Indicate if the mentioned expression yields an eigenvalue equation, and if so, indicate the eigenvalue. (x(2/a)1/2 sin2piex/a)
- Indicate if the mentioned expression yields an eigenvalue equation and if so, indicate the eigenvalue. (-h2/2md2/dx2 +0.5) sin2piex/3
- Indicate if the mentioned expression yields an eigenvalue equation and if so, indicate the eigenvalue. d/dy(e^-y2)
- Indicate if the mentioned expression yields an eigenvalue equation, and if so, indicate the eigenvalue. 3(4lnx2) 3^=3x
- At what position would you expect to observe IR absorptions for the given molecule?
- At what position would you expect to observe IR absorptions for the given molecule below?
- Determine the number of IR-active vibrations for the following molecule. Determine their symmetry first. Formaldehyde, H_2CO
- Indicate if the mentioned expression yields an eigenvalue equation and if so indicate the eigenvalue. -ihd/dxe^imx, where m is a constant
- Find the number of normal mode vibrations observed for the given molecule. N2O
- The major component of clove oil will exhibit a large, broad peak centered around 3500 cm-1 in an IR spectrum. What functional group will give rise to such a band in the IR spectrum?
- Question 2 Identify the signals you would expect in the diagnostic region of the IR spectrum for the following compound. C-H stretching signals just below 3000 cm-1, one signal around 1720 cm-1 and o
- The approximate positions for which the following compounds show IR absorptions is
- Which of the biphenyls are optically active options are there in image?
- Name two regions on the IR spectra.
- 1. Name two regions on the ir spectra
- The IR C=C stretching absorption in symmetrical alkynes is usually absent. Why?
- IR spectroscopy can often be used to eliminate possible compounds because of the lack of IR absorptions expected for key functional groups. Given the following IR data, which functional group(s) can
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d/dx(e^x)
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. -ihd/dxsinpiex/2
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d/dx sinpiex/2
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d/dxcos4x
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. px(sin2piex/3)
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d2/dx2cos4piex
- Indicate if the given expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d2/dx2 sinpiex/2
- Determine the number of IR-active vibrations for the following molecule. Determine their symmetry first. Oxalic acid, (COOH)_2
- The approximate positions at which the following compounds show IR absorptions?
- Indicate if the depicted expression yields an eigenvalue equation, and if so, indicate the eigenvalue. d2/dtheta2 sinpie costheta
- Reduction of d-glucose leads to an optically active alditol (d-glucitol), whereas reduction of d-galactose leads to an optically inactive alditol. Explain.
- What types of vibrations are encountered in organic molecules?
- At what position would you expect to observe IR absorptions for the following molecule?
- At what position would you expect to observe IR absorptions for the following molecule below?
- Identify the mentioned aldose, and indicate whether the given aldose is a D or L sugar.
- 1) Under C4v symmetry, which of the following irreducible representations, if any, correspond to a Raman active vibration? A1,A2,B1,B2,E 2) Under C4v symmetry, which of the following irreducible rep
- Specify the types of strain that destabilize the conformer shown below. 1.Torsional strain ---- 2.Angle (ring) strain ------ 3.Steric (van der Waals) strain----- [{Image src='bonds4653120621465388925