2-Butene-1-thiol is one component of skunk spray. How would you synthesize this substance from...
Question:
2-Butene-1-thiol is one component of skunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene?
Skunk Spray:
Skunk spray is a yellow oil that is highly volatile, meaning it quickly evaporates and turns into a gas. Skunk spray contains organic sulfur-containing thiols, which give it its distinctive odour. The odour of sulphur is usually attributed to that rotten eggs.
Answer and Explanation: 1
Become a Study.com member to unlock this answer! Create your account
View this answerSynthesis of 2-butene-1-thiol from methyl-2-butenoate:
As shown below, 2-butene-1-thiol can be synthesized from methyl-2-butenoate:
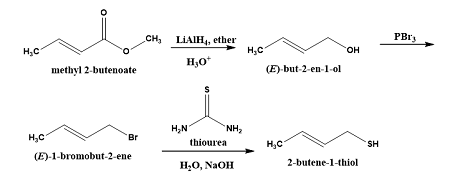 |
The ester...
See full answer below.
Ask a question
Our experts can answer your tough homework and study questions.
Ask a question Ask a questionSearch Answers
Learn more about this topic:

from
Chapter 15 / Lesson 21Synthetic organic chemicals are man-made substances containing carbon atoms that are not naturally occurring. Explore the differences between natural and synthetic organic chemicals through examples such as those found in plastics, medicine, and sweeteners.
Related to this Question
- The drug cocaine hydrochloride hydrolyzes in air to give methyl benzoate. Its odor, which smells like pineapple guava, is used to train detection dogs. (14.3) a. Draw the condensed structural formula
- Sulfur ylides, like phosphorus ylides, are useful intermediates in organic synthesis. Methyl trans-chrysanthemate, an intermediate in the synthesis of the insecticide pyrethrin I, can be prepared from diene and a sulfur ylide. Draw a stepwise mechanism fo
- Sulfur-containing compounds give skunks their potent smell. One of the principal smelly compounds in skunk spray is (E)-2-butene-1-thiol, C4H7S. (a) What is the root-mean-square (rms) molecular speed of a gas molecule of this compound at 25 C? (b) Using
- Following is the structural formula of musk ambrette, a synthetic musk, essential in perfumes to enhance and retain odor. Propose a synthesis of this compound from m-cresol (3-methylphenol).
- An organic molecule that must bind to an enzyme to make it function properly is a(n): a) apoenzyme. b) holoenzyme. c) isomerase. d) inhibitor. e) coenzyme.
- The compound MON-0585 is a nontoxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromatic rings.
- Using any two of lysozyme, carboxypeptidase, chymotrypsin or aspartate transcarbamoylase, concisely explain the mechanism of catalysis in detail.
- Entacapone is a component of a drug used to treat Parkinson's disease. [Table] The acidic protons in entacapone are the ones on the OH groups. Which is the most acidic? Support your explanation using chemical structures.
- Is the biochemical pathway responsible for the greatest portion of drug metabolism? a) Therapeutic Drug Monitoring b) Toxicology c) Endocrinology d) Mixed Function Oxidase (MFO) System
- Entacapone is a component of a drug used to treat Parkinson's disease. The acidic protons in entacapone are the ones on the OH groups. Which is the most acidic? Support your explanation using chemical structures.
- In addition to methyl groups, tetrahydrofolate (THF) can carry formaldehyde groups that are important for the biosynthesis of thymidine. Draw a detailed mechanism for incorporation of the formaldehyde into THF to give the following product.
- A compound known as mature is the sex pheromone of the pine bat scale and it was synthesized in several steps from alcohol citronellol. A key intermediate in the synthesis was the nitrile (S)-4,8-dime
- Give the structure of: a. trans-cinnamic acid b. urea c. naphthalene
- What are the two heterocyclic compounds in quinine? A) pyridine B) quinoline C) pyrrole D) styrene
- The PDB page for 4DXH indicates that horse liver alcohol dehydrogenase is complexed to the cofactor ______ and ______ form of the substrate. a) NAD+ and unreactive. b) NAD+ and reactive. c) NADH and unreactive. d) NADH and reactive.
- PGF2 alpha is synthesized in cells from arachidonic acid (C20H32O2) using a cyclooxygenase enzyme that catalyzes a multistep radical pathway. Part of this process involves the conversion of radical A to PGG2, an unstable intermediate, which is then transf
- Irradiation of poly(1,3-butadiene) followed by addition of styrene yields a graft copolymer that is used to make rubber soles for shoes. Draw the structure of a representative segment of this styrene-butadiene graft copolymer.
- Which of the following is used as a disinfectant but is a potentially lethal protoplasmic poison that can be absorbed through the skin? a) Chloroform b) Cresol c) Phenol d) Hexachlorophene
- Coniine, C_8H_17N, is the toxic principle of the poison hemlock drunk by Socrates. When subjected to Hofmann elimination, coniine yields 5-(N,N-dimethylamino)-1-octene. If coniine is a secondary amine, what is its structure?
- Entacapone is a component of a drug used to treat Parkinson's disease. The acidic protons in entacapone are the ones in the OH groups. Which is the most acidic?
- Explain how you would synthesize a fully head-to-head analog of polypropylene by radical polymerization. Draw the structure of the monomer, and any relevant intermediates in the synthesis.
- \alpha -Fenchone is a pleasant-smelling terpenoid isolated from oil of lavender. Propose a pathway for the formation of a-fenchone from geranyl diphosphate. A carbocation rearrangement is required.
- Ephedrine, a central nervous system stimulant, is used in nasal sprays as a decongestant. This compound is a weak organic base: C10H15ON (aq) + H2O (l) leftrightharpoons C10H15ONH+ (aq) + OH- (aq) A
- It is the biochemical pathway responsible for the greatest portion of drug metabolism. a) Therapeutic Drug Monitoring. b) Toxicology. c) Endocrinology. d) Mixed Function Oxidase (MFO) System.
- A small ion that must bind to an enzyme to make it function properly is a(n): a) apoenzyme. b) coenzyme. c) cofactor. d) inhibitor. e) holoenzyme.
- 1-Butanethiol (CH3CH2CH2CH2SH) is synthesized by reacting thiol radical (HS ) with butene in the presence of hydrogen disulfide (HS- SH). The HS radical is formed by shining UV light on HS-SH. It (1-butanethiol) is added to stink bombs because it has a v
- Valganciclovir, marketed as Valcyte, is an antiviral agent used for the treatment of cytomegalovirus. Called a prodrug, valganciclovir is inactive by itself but is rapidly converted in the intestine by hydrolysis of its ester bond to produce- How might va
- The molecule cyanazine, once used as a herbicide, has now been phased out by the EPA because of its significant human toxicity. a. Add lone pairs, as needed, to the structure of cyanazine. b. Indicate
- What residue in Cytochrome c oxidase participates in forming a transient radical?
- 1,6-Hexanediamine, a starting material needed for making nylon, can be made from 1,3-butadiene. How would you accomplish the synthesis? H_2C=CHCH=CH_2overset{?}{rightarrow} H_2NCH_2CH_2CH_2CH_2CH_2CH_2NH_2
- Iodoacetate is an irreversible inhibitor of GAPDH. Why? How would you experimentally demonstrate that it targets the catalytic residue of the protein?
- Identify all the compounds that are naturally found in your body. \\ 1. Norepinephrine 2. Testosterone 3. Caffeine 4. Guanosine 5. Uridine 6. Artemisinin
- Write a mechanism for the conversion of alpha-ketoglutarate to succinyl CoA in step 4 of the citric acid cycle.
- N, N-Diethyl-m-toluamide (DEET) is the active ingredient in many insect-repellent preparations. How might you synthesize this substance from m-bromotoluene?
- Which of the following odors does the ester, methyl salicylate, produce? a) cinnamon b) avocado c) orange d) oil of wintergreen
- Moxisylyte, an alpha-adrenergic blocker, is used as a peripheral vasodilator. Propose a synthesis for this compound from thymol, which occurs in the volatile oils of members of the thyme family. Thymol is made industrially
- Identify all substances that can be classified as alkaloids. \\ a. ciprofloxacin b. uridine c. aflatoxin B1 d. norepinephrine e. cocaine f. tetrodotoxin
- What can happen if a carboxylic acid ester (e.g. methyl caproate) is exposed to a slightly acidic aqueous environment (e.g. sweaty skin) for a period of hours? The fruity pineapple odor of methyl caproate would change to what kind of odor?
- What is the main chemical constituent of the oil of cardamom that is responsible for the flavor of this oil? A. Cineole B. Engenol C. Geraniol D. Limonene
- Treating trimethylamine with 2-chloroethyl acetate gives acetylcholine as its chloride. Acetylcholine is a neurotransmitter. Propose a structural formula for this quaternary ammonium salt and a mechanism for its formation.
- What is a thiol (what functional group determines a thiol) and what is its distinctive characteristic?
- Two molecules used in the polymer industry are azodicarbonamide and methyl cyanoacrylate. Their structures are: Azodicarbonamide is used in forming polystyrene. When added to the molten plastic, it decomposes to nitrogen, carbon monoxide, and ammonia gase
- Laboratory 11B: Methyl Orange 1) What changes to the synthetic sequence used for methyl orange would need to be made to generate FD&C Orange #1 (see lab manual for the structure). (Some structure dra
- Brevicomin, the aggregation pheromone of the western pine bark beetle, contains a bicyclic bridged ring system and is prepared by the acid-catalyzed cyclization of 6,7-dihydroxy-nonan-2-one. a. Suggest a structure for brevicomin. b. Devise a synthesis o
- Beeswax is a wax created by bees when they make their honeycomb. Its major component is triacontanyl palmitate which is an ester of the alcohol triacontanol, HO-(CH2)29CH3, and palmitic acid. Using line-angle formulas draw the structure of beeswax.
- Acid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?
- Mitomycin is a naturally occurring molecule that is effective at killing cancerous cells. Classify the amines in mitomycin as primary, secondary, and tertiary. The carbamate group (NH2O2) of mitomycin is different from an amine because it has different ch
- An inorganic substance that assists in an enzyme catalytic process is called a: a. cofactor b. coenzyme c. regulator d. substrate
- Identify all compounds that can be classified as phenols. \\ 1. norepinephrine 2. testosterone 3. turmeric 4. a-tocopherol 5. fluorescein 6. morphine
- Devise a synthesis of (E)-tetradec-11-enal, a sex pheromone of the spruce budworm, a pest that destroys fir and spruce forests, from acetylene, Br(CH_2)_{10}OH, and any needed organic compounds or inorganic reagents.
- 3) The pyridoxal phosphate-dependent enzyme, Histidine decarboxylase, catalyzes the decarboxylation of the amino acid histidine in the biosynthesis of the bioactive, small molecule histamine. Draw sturctures and use two headed arrows to illustrate the ste
- Is DMSO a protic or aprotic solvent?
- Show two methods for the synthesis of dopamine, a neurotransmitter involved in regulation of the central nervous system. Use any alkyl halide needed.
- Acetylcholine is a neurotransmitter that transmits a signal from a nerve cell to a muscle cell, causing the muscle to contract. How is the neurotransmitter inactivated so that the muscle can relax? a. It is denatured by the local pH and loses its active
- Arylamines and heterocyclic aromatic amines are considerably less basic than alkylamines. Justify.
- What is the name of the ester responsible for the flavor and odor of an orange?
- Propose a mechanistic pathway for the biosynthesis of caryophyllene, a substance found in clove oil.
- p-Aminobenzoic acid (PABA) is widely used as a sunscreen agent. Propose a synthesis of PABA starting from toluene.
- Which proton of the following drug is most acidic? THC is the active component in marijuana, and ketoprofen is an anti-inflammatory agent.
- Which proton in the following drug below is most acidic? THC is the active component in marijuana, and ketoprofen is an anti-inflammatory agent.
- ABA type of triblock copolymer Where A and B represent methyl methacrylate (MMA) and styrene (St) respectively. Please note that a living PSt anion can initiate MMA, but a living PMMA anion cannot initiate St.Explain?
- Serine residue is often the site of covalent inhibition. Why?
- How do the mechanisms of base-catalyzed enolization and acid-catalyzed enolization differ?
- ________ is taken up by cells using receptor-mediated endocytosis, which involves the recognition of the B-100 apolipoprotein component of LDL.
- Outline a synthetic route to convert methylenecyclohexane into cyclohexylacetic acid.
- Serine can be synthesized by a simple variation of the amidomalonate method using formaldehyde rather than an alkyl halide. How might this be done?
- There are poly-ether-urethanes and poly-ester-urethanes. Which one breaks down more easily in the biological environment of the human body?
- The use of phenols such as tert-butylcatechol as free-radical scavengers is based on the fact that phenolic hydrogens are generally abstracted by radicals, producing relatively stable phenoxyl radicals that interrupt chain processes of oxidation and polym
- A butadiene styrene is made by polymerizing 2 monomers of styrene with 6 monomers of butadiene. If 22% of the crosslink sites are to be bonded with sulfur, what wt.% sulfur is required?
- The following processes result in a stronger polystyrene polymer. Explain why in each case. a. addition of catalyst to form syndiotactic polystyrene b. addition of 1,3-butadiene and sulfur c. producing long chains of polystyrene d. addition of a catal
- What is the efficient synthesis mechanism of the following compound known as muscalure, the sex pheromone of the common housefly?
- Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1,3-butadiene. How would you carry out this synthesis? H_2C = CHCH = CH_2 \rightarrow^{3 steps} N \equiv CCH_2CH_2CH_2CH_2C \equiv N
- The cofactor that is bound to the enzyme at all times is called? A) Coenzyme B) Prosthetic Group c) Holoenzyme d) Metal ion e) Apoenzyme
- Amphetamine is a powerful stimulant of the central nervous system. (a) Which proton in amphetamine is most acidic? (b) What products are formed when amphetamine is treated with NaH? (c) What products are formed when amphetamine is treated with HCl?
- If Malonyl-CoA is synthesized from Acetyl-CoA and ^{14}CO_2 where will the ^{14}C end appear in palmitic acid?
- Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not. Explain.
- How does the conversion of L-alanine to D-alanine occur through the action of alanine racemase, a PLP-dependent isomerase, and why is D-alanine important as a constituent of peptidoglycan, the building material of bacterial cell walls?
- What is the mechanism for the key Dieckmann condensation step used in the synthesis of strychnine by R.B. Woodward, which played a significant role in the development of synthetic planning and execution?
- Glutamic acid, a non-essential amino acid, is synthesized from a number of amino acids in the body. The structural formula of glutamic acid is Write the fully ionized form of the molecule.
- An anticancer drug with oxirane moiety is
- What is the mechanism of action of dantrolene? A) MAO inhibitor B) Dopamine receptor agonist C) Antagonist at RYR1 D) Agonist at GABAB receptors
- The herbicide atrazine binds to the photosynthetic apparatus and in the process is anchored by two critical protein residues: a phenylalanine and a serine. (a) Describe three interactions that occur between these residues and the herbicide. (b) Over tim
- Discuss two methods to prepare crosslinked poly(methyl methacrylate (PMMA). Sketch chemical structures when needed.
- What is the structural formula for Pectic acid, the main component of pectin, which is responsible for the formation of jellies from fruits and berries? Pectic acid is a polymer of D-galacturonic acid joined by alpha-1, 4-glycosidic bonds.
- Apomorphine, bromocriptine, and ropinirole all share which mechanism of action? a. Blocking central muscarinic receptors b. Agonist dopamine receptors c. Alpha-2 adrenoceptor agonists d. Antagonist dopamine receptors
- In order to create an engineered human hemoglobin molecule that had the same bicarbonate binding properties as crocodile hemoglobin, twelve amino acid residues had to be changed. Not all of these resi
- Cocaethylene is a metabolite of cocaine that can be created under particular circumstances. Describe the in-vivo synthesis of Cocaethylene in humans.
- A gamma-hydroxyaldehyde may cyclize to form: A) a hemiacetal. B) an ester. C) a pyrimidine. D) a lactone. E) a lactam.
- During Edman degradation, N-terminal amino acids first undergo a chemical reaction and then they are (a) identified by trypsin digest. (b) extracted into organic solvent. (c) dansylated and identified by chromatography. (d) cleaved off by an exopeptid
- Identify the substance(s) used in medicine as anesthetics. \\ 1. Caffeine 2. Lidocaine 3. Sucralose 4. Zoloft 5. Cocaine 6. Methyl jasmonate
- Valganciclovir, marketed as Valcyte, is an antiviral agent used for the treatment of cytomegalovirus. Called a prodrug, valganciclovir is inactive by itself but is rapidly converted in the intestine by hydrolysis of its ester bond to produce- What atoms p
- Valganciclovir, marketed as Valcyte, is an antiviral agent used for the treatment of cytomegalovirus. Called a prodrug, valganciclovir is inactive by itself but is rapidly converted in the intestine by hydrolysis of its ester bond to produce- What role do
- When a patient has accidentally ingested methanol, a physician can order the use of ethanol to overwhelm the alcohol dehydrogenase enzyme. This prevents the liver from producing deadly formaldehyde from the methanol. This is an example of: (a) covalent m
- When 5-Fluorouracil interacts with its target enzyme and Methylene Tetrahydrofolate, it forms non-hydrolyzable covalent bonds linking all 3 molecules together. What type of inhibitor is 5-fluorouracil? A. Competitive B. Mixed C. Activator D. Allosteric E.
- Dimethyl sulfide is the most abundant biological sulfur compound emitted into the atmosphere. It is produced by phytoplankton and emitted over the oceans. Draw the structure of dimethyl sulfide.
- Tris(hydroxymethyl)aminomethane or (HOCH_2)_3CNH_2, commonly called TRIS or Trizma, is often used as a buffer in biochemical studies. Its buffering range is from pH 7 to 9, and K_b is 1.19\times 10^(-6) for the reaction: (HOCH_2)_3CNH_2(aq)+H_2O(l)\rightl
- What is the known human carcinogen that is used when making polychloroprene?
- Phosphoglycerides containing choline as the amino alcohol unit are called: a) cephalins b) cardiolipins c) sphingomyelins d) citicolines e) lecithins
- The mosquito attractant oct-1-en-3-ol can be prepared by the reduction of oct-1-en-3-one. How can UV spectroscopy be used to monitor this reaction?
- 1-Octen-3-ol, a potent mosquito attractant commonly used in mosquito traps, can be prepared in two steps from hexanal, CH_3CH_2CH_2CH_2CH_2CHO. What is the structure of the product from the first step, and how can it be converted into 1-octen-3-ol?